Vertically Stacked High Mobility Channel Transistors

- ENGINEERING & TECHNOLOGIES
- Text & Image
- November 24,2020

Taiwan has officially become an aging society with more than 14% of the population over 65 years old in 2018. It is estimated that Taiwan will become a super-aged society in 2026, during which there will be over 20% of the total population being elderly. Meanwhile, many countries around the world are also facing the challenge of society aging. These challenges and the needs for telemedicine and mobile healthcare have driven the research and development of smart medicine and precision health.
The WHO defined eHealth (smart medicine) as “the use of information and communication technologies (ICT) for health,” which includes medical healthcare, disease control, public health, research and education. The current development of smart medicine aims to solve the problem of limited medical staff and build high quality medical services by cross-disciplinary research and integration of resources.
Cross-disciplinary Development of Smart Medicine
The research and development of smart medicine rely on the ICT and biomedical industry as well as Internet technology, sensor chips manufacture, software development, data analysis, and medical device development. In order to analyze the research trend of smart medicine, journal papers with keywords “Smart Medicine”, “eHealth”, Smart Healthcare”, “Smart Hospital”, “Smart Health”, and “Mobile Health” were retrieved from the Web of Science database. The result indicates that the top three research fields related to smart medicine are Healthcare Sciences & Services, Medical Informatics, and Public, Environment & Occupational Health, following by Computer Science & Information Systems, and Physiatry (Fig.1).
The operation of the medical system can be improved with the integration of a wide variety of smart medicine technologies. Medical decisions, treatments, practices could be supported by implementing the advanced Internet system, AI technology, and big data to medical imaging and wearable medical devices. Altogether, these smart medicine technologies can help alleviate the workload of health care staff and provide higher quality and efficiency medical services.
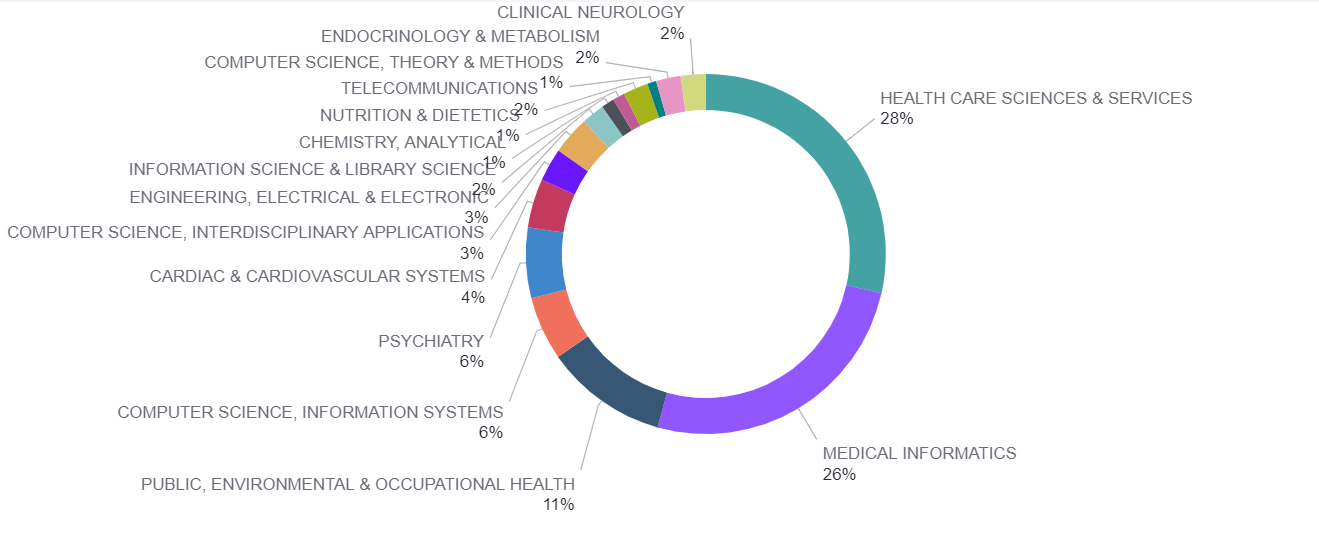
Fig1. Research categories of global smart medicine-related journal papers from 2016 to 2020 (Resources: InCites)
International Smart Medicine Research Trend
Many countries have been developing smart medicine and digital health due to falling birth rates and the aging population. Taking a closer look at the smart medicine papers publication trend (Fig.2), it is shown that the publication amount has increased by over 100% (from 438 pieces of papers to 946) from 2016 to 2019. Meanwhile, the paper published in 2020 as of present has reached 745 pieces, which is more than the year of 2016 to 2018. In 2020, the pandemic has brought challenges to medical systems in many countries; however, it has also driven the development of smart medicine and its cross-disciplinary application, such as using ICT to develop telemedicine.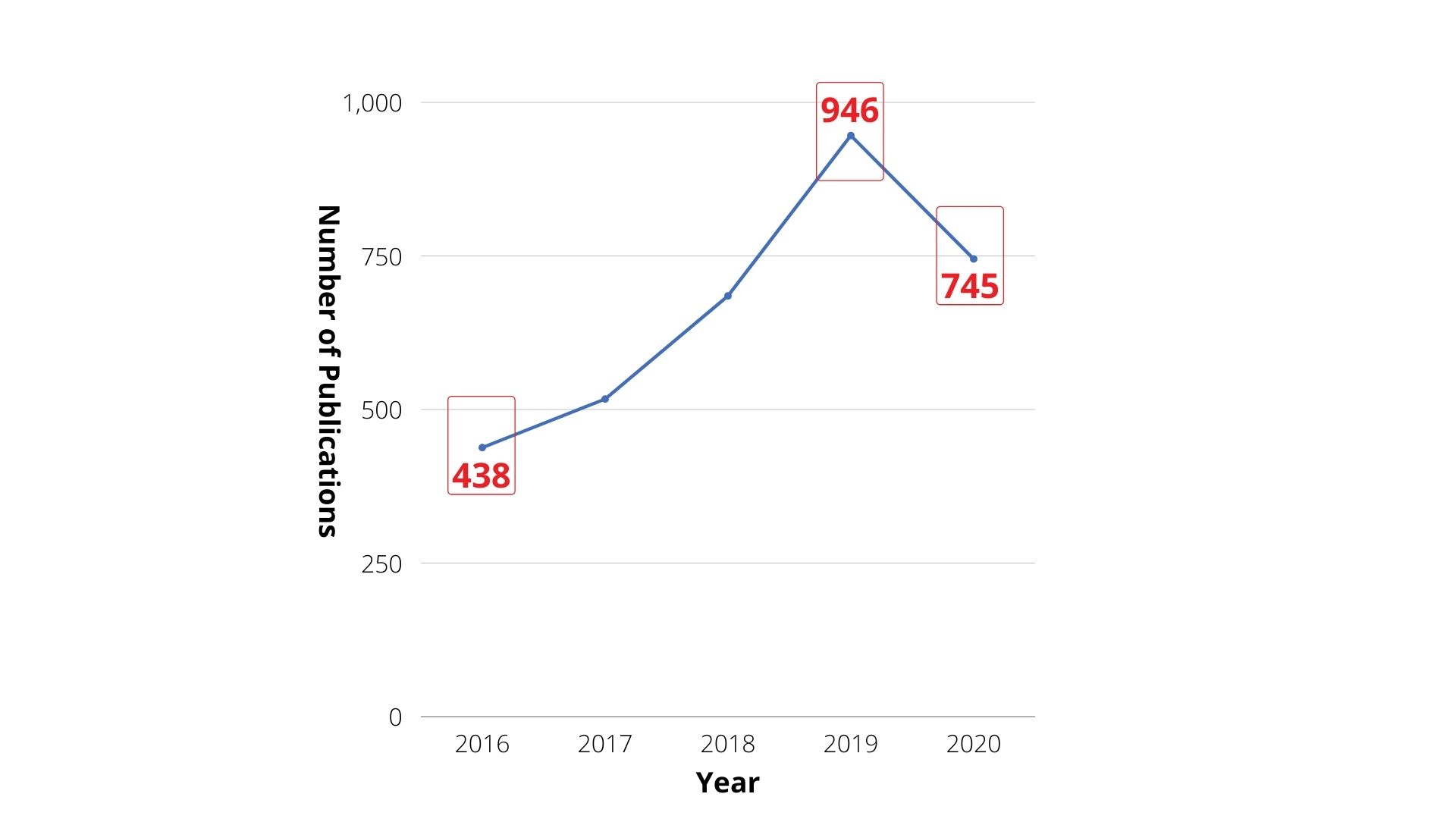
Fig2. International smart medicine paper publication trend from 2016 to 2020 (Resources: InCites)
Comparing the quantity of different countries’ smart medicine paper publication, the U.S. ranked first with 1698 pieces of papers, following by the U.K. (332 pieces) and Australia (261 pieces). Among all countries in the database, Taiwan ranked 21st with 44 publications. (Fig.3)
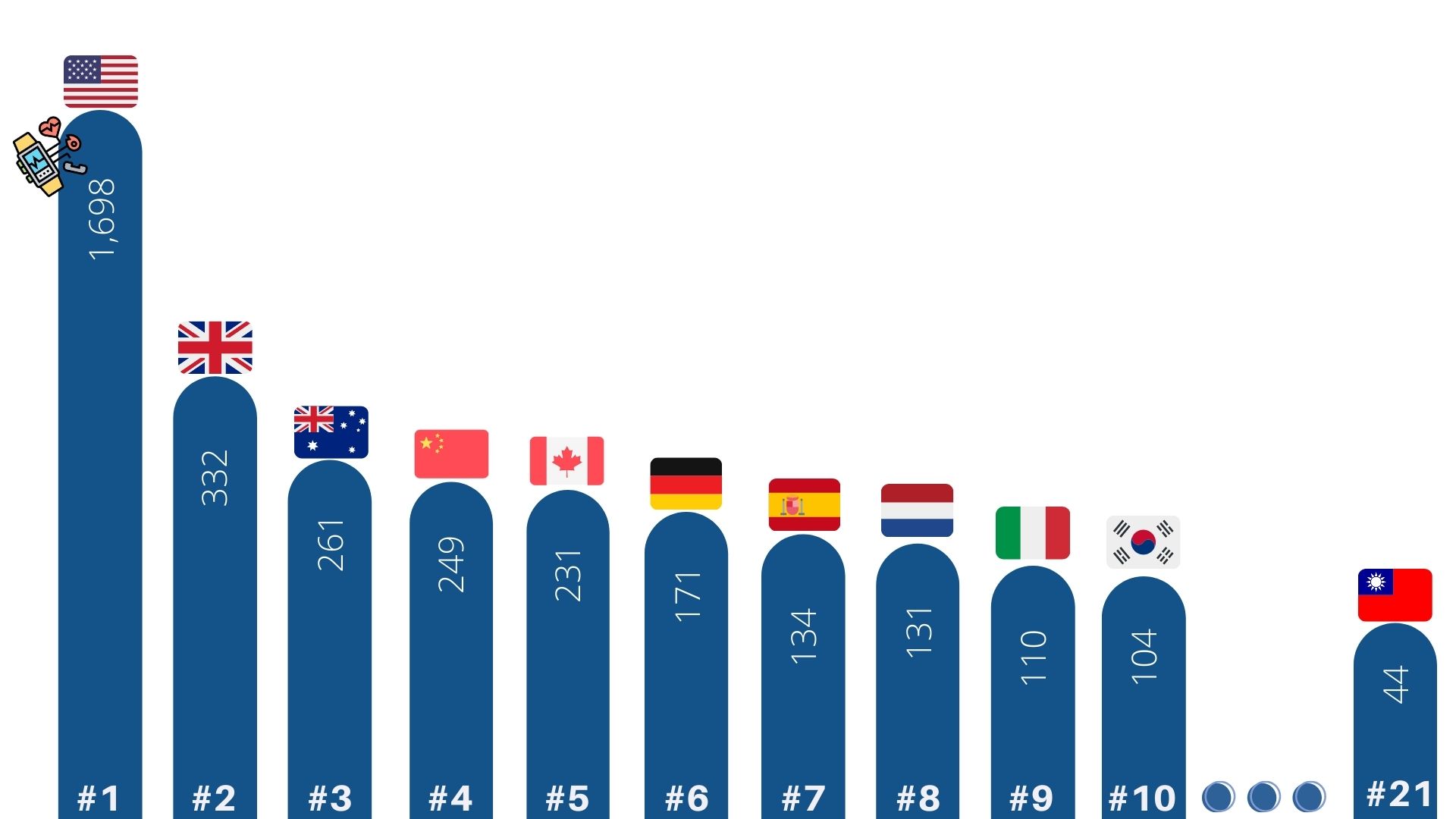
Fig 3. Global smart medicine paper publication numbers and ranking from 2016 to 2020 (Resources: InCites)
Countries around the world have also implemented policies and strategies to boost the research of smart medicine. Take the U.S., the leading country of current smart medicine development, as an example, the U.S. FDA proposed “Digital Health Innovation Action Plan” to focus on the development of mobile medical applications, medical device data systems, portable medical devices, telemedicine, and personalized healthcare. Meanwhile, the EU is also actively developing smart medicine technologies, especially focusing on personal health data protection. MyHealthMyData (MHMD), a Horizon 2020 Research and Innovation Action, aims at fundamentally changing the way sensitive data are shared through pioneering blockchain and medical informatics technologies.
As in Asia, South Korea activated a blockchain-based smart hospital project to improve the medical service platform and build a comprehensive medical information platform for automatic and personalized services, such as providing prescriptions and applying for medical insurance.
Taiwan’s Opportunities in Smart Medicine Development
Taiwan has a strong ICT industry background, a comprehensive health insurance system, and an extraordinary medical system. Altogether with the government’s support, the country has a solid foundation for the development of smart medicine. Take AI medical imaging as an example, MOST proposed a medical imaging project in 2017 that aims at building an AI medical imaging database and has established Taiwan’s first “AI medical imaging database,” which recorded 15 types of diseases and more than forty thousand of image data. The database is open to research teams in Taiwan to develop a better AI medical imaging recognition assisting system.
To address the problems of an aging society, Taiwan should continue to combine the country’s R&D strengths to develop a more comprehensive and competitive smart medicine industry. However, we should also acknowledge that smart medicine is not just an outcome of the integration of ICT and medicine, but also the accelerating development of novel medical technologies that bring medical services to a brand-new era. As a result, the government should take medical ethics and personal data protection into account and implement regulations and laws when developing smart medicine at full speed.
STAY CONNECTED. SUBSCRIBE TO OUR NEWSLETTER.
Add your information below to receive daily updates.




