Transmembrane and Coiled-Coil Domain 1 Impairs the AKT Signaling Pathway in Urinary Bladder Urothelial Carcinoma: A Character...

Author(s)
Chi-Tai Yeh, Alexander T. H. Wu, Zhen Yang, Yew-Min TzengBiography
Dr. Yew-Min Tzeng is a Professor of Biochemical Engineering and the President of National Taitung University. Dr. Tzeng was granted a Ph.D. from the University of Toronto in Chemical Engineering and Applied Chemistry in 1987. Dr. Tzeng’s research interests are in bioprocess engineering, bio-pesticides, food biotechnology, and natural herbal products.
Academy/University/Organization
National Taitung University-
TAGS
-
Share this article
You are free to share this article under the Attribution 4.0 International license
- LIFE SCIENCES
- Text & Image
- July 20,2020
Compounds derived from natural resources and advancements in synthetic chemistry have become the main avenue for the development of cancer therapeutics. Representative compounds derived from terrestrial plants such as taxol, vincristine, and topotecan have been widely used as anti-cancer agents. Taiwan is geologically gifted in terms of biodiversity and is a treasure island of natural plantations. Antrodia cinnamomea (AC), a fungus species exclusively found in Taiwan and traditionally used by the indigenous people for a variety of disorders, represents an ideal source for therapeutic discovery. Over the past two decades, President Tzeng’s team and collaborators have focused on the isolation, purification and characterization of the multiple bioactive compounds from AC. One of the ubiquinone derivatives, 4-Acetylantroquinonol B (4-AAQB), has been characterized as having numerous biological and pharmacological properties, including anti-cancer agents. Based on the growing demand for cancer therapeutics, 4-AAQB represents a potential lead for Taiwan’s entry into the global pharmaceutical supplier arena. Further development of 4-AAQB will require institutional support in sustainable biopharmaceuticals, chemical biology, and clinical medicine, all of which will contribute to Taiwan’s advancement in green biotechnology. Thus, 4-AAQB and AC natural compounds may be a hidden jade waiting to be discovered.
During the past four decades, cancer has consistently ranked as the top cause of death in Taiwan. The socioeconomic impact on families affected by cancer is unimaginable. Despite the development of new drugs and the improvement in medical technologies, cancer patients continue to go through different therapeutic modalities and endure associated physical and mental agonies before succumbing to the disease. Recently, compounds and/or extracts derived from natural resources have gained much interest for developing new candidates of therapeutic and supplementing purposes. Accumulating evidence indicates that natural compounds of different origins contain a spectrum of biological and pharmacological properties in both cellular and preclinical animal experimental models. Notably, studies have demonstrated that natural compounds can also inhibit tumor growth as effectively as clinically available chemotherapeutic agents, but with a significantly higher tolerance and reduced side effects. Antrodia cinnamomea (AC) is a rare mushroom exclusively found in Taiwan’s high mountains (Figure 1). AC has long been regarded as the treasure of Taiwan's cloud forests by indigenous peoples due to its potent effects and efficacy in treating a wide range of illnesses, and in promoting health. It was not until recent years that emerging studies demonstrated that compounds derived from AC also contain anti-cancer properties1. Research teams led by Professor Tzeng, Professor Yang from Peking University, and collaborators from Shuang-Ho Hospital and Taipei Medical University collectively isolated and characterized an active ubiquinone derivative from AC, 4-Acetylantroquinonol B (4-AAQB) with anti-cancer functions in recent years (Figure 2).

Figure 1. Fruiting Body of Antrodia cinnamomea (J Nat Prod 2014 77:118)
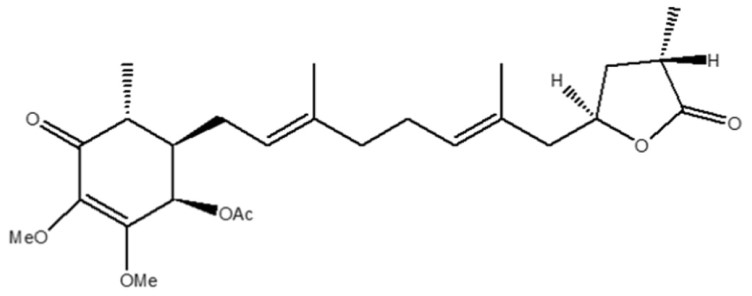
Figure 2. 4-Acetylantroquinonol B (4-AAQB)
Professor Tzeng’s team has dedicated their efforts to characterizing 4-AAQB as a lead anti-cancer compound for different cancer types (Table 1). Their research results have been published in international peer-reviewed journals such as Toxicology and Applied Pharmacology and Cancers. 4-AAQB appears to downregulate major oncogenic signaling pathways, such as Lgr5/Wnt/β-catenin, JAK-STAT, and non-receptor tyrosine kinases (nRTKs), and induces the expression of the small molecule ribonucleic acid has-miR-324 in colon cancer. In addition, 4-AAQB enhanced the sensitivity of FOLFOX-resistant colon cancer cells in preclinical models2,3. In ovarian cancer cells, 4-AAQB treatment led to the suppression of PI3K/Akt/mTOR/p70S6K signaling, the inhibition of Atg-5-mediated autophagy, self-renewal ability and sensitization to cisplatin4. Furthermore, 4-AAQB appears to block β-catenin/ TCF-1/STAT3 signaling pathways (Figure 3) and significantly inhibits the growth of malignant glioblastoma multiforme (GBM) both in vitro and in vivo5. Our most recent findings showed that 4-AAQB treatment led to cell cycle arrest at the G0/G1 stage in triple-negative breast cancer (TNBC) cells, with an increased expression of p21 and p27, and a decreased level of CDKs 2, 4, and 6, Cyclins A, D and E, BRCA1 and pCHEK1/2 proteins6. Collectively, this evidence indicates that AC represents a great treasure for the discovery of pharmacologically and functionally active compounds, as exemplified by 4-AAQB.
Table 1. Oncogenic Signaling Targeted by 4-AAQB in Different Cancer Types
| Cancer Type | affected Genes | Reference |
|---|---|---|
| Colorectal | Lgr5, β-catenin, p-Jak2, p-STAT3, p-Src, SOD2, hsa-miR-324-5p | 2,4 |
| Ovarian | Atg-5, Atg-7, PI3K, AKT, mTOR, p70S6K | 3 |
| GBM | β-catenin, LEF1, p-STAT3, p-GSK-3β | 5 |
| Triple-Negative Breast Cancer (TNBC) | CDK2、CDK6、CDK4、Cyclin D、Cyclin E、Cyclin A、BRCA1 and pCHEK1/2 | 6 |
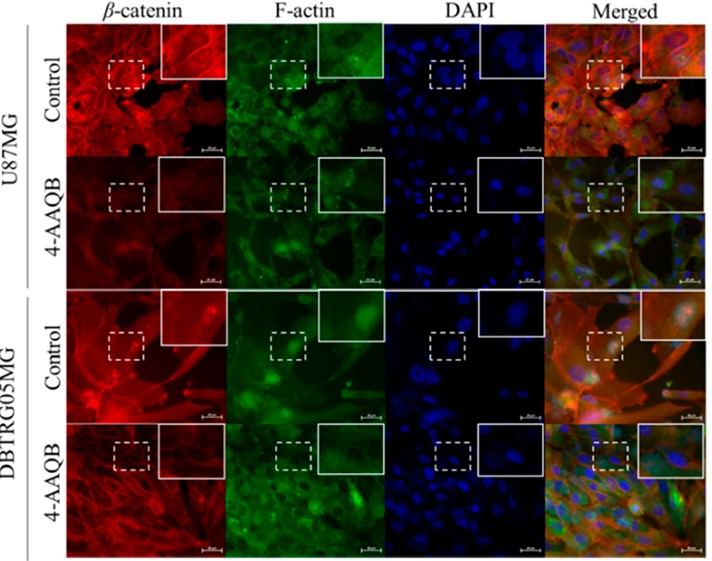
Figure 3. Immunofluorescence Staining Showed 4-AAQB Inhibits a Powerful oncogene, β-catenin in GBM cells (Cancers 2018 10:491)
Leading with therapeutic potential of 4-AAQB in cancer (Figure 4), Professor Tzeng has teamed up with Professor Zhen Yang, from the College of Chemistry and Molecular Engineering, Peking University, to characterize functional compounds and bio-active metabolites derived from AC. Subsequently, a pipeline of AC derived parent compounds and chemically synthesized derivatives have been established by this collaborative effort. In closing, 4-AAQB and serial compounds isolated from Antrodia cinnamomea have demonstrated a tremendous value and have shone a light on Taiwan’s potential contribution to innovation and sustainable development on the world stage.
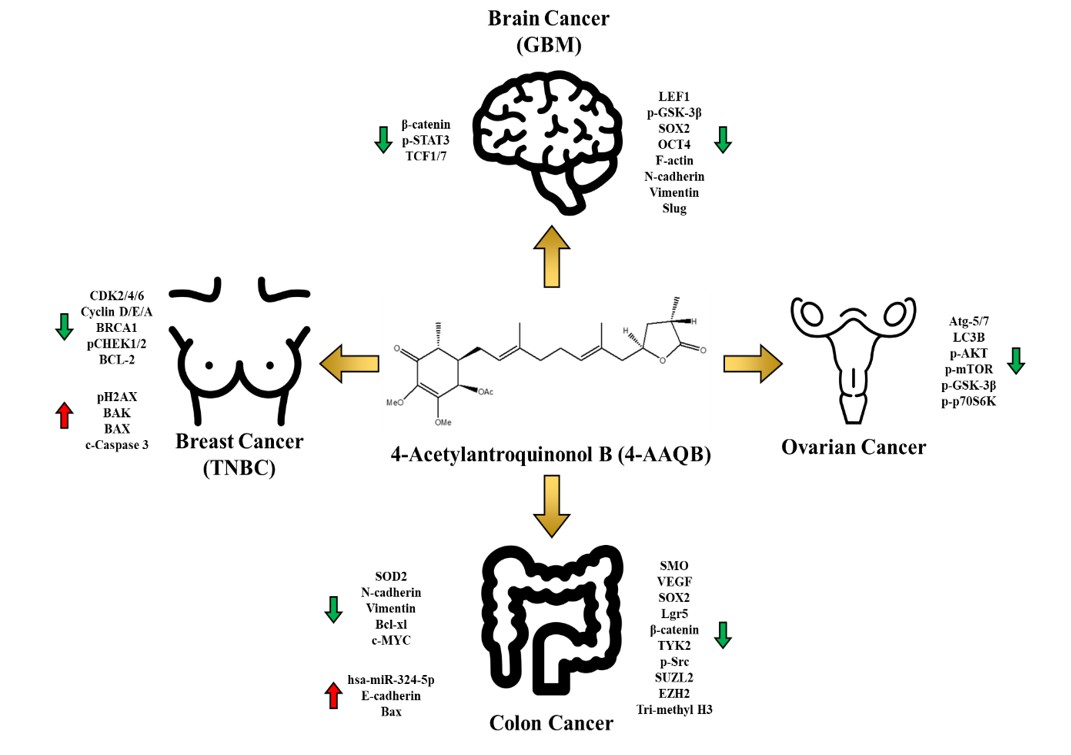
Figure 4. Summary of Therapeutic Potential of 4-AAQB
Reference:
- Geethangili M, Tzeng YM. Review of Pharmacological Effects of Antrodia camphorata and Its Bioactive Compounds. Evid Based Complement Alternat Med. 2011;2011:212641.
- Chang TC, Yeh CT, Adebayo BO, Lin YC, Deng L, Rao YK, Huang CC, Lee WH, Wu AT, Hsiao M, Wu CH, Wang LS, Tzeng YM. 4-Acetylantroquinonol B inhibits colorectal cancer tumorigenesis and suppresses cancer stem-like phenotype. Toxicol Appl Pharmacol. 2015 Oct 15;288(2):258-68.
- Bamodu OA, Yang CK, Cheng WH, Tzeng DTW, Kuo KT, Huang CC, Deng L, Hsiao M, Lee WH, Yeh CT. 4-Acetylantroquinonol B suppresses SOD2-Enhanced cancer stem cell-like phenotypes and chemoresistance of colorectal cancer cells by inducing hsa-miR-324 re-expression. Cancers (Basel). 2018 Aug 10;10(8):269.
- Liu M, Bamodu OA, Huang WC, Zucha MA, Lin YK, Wu ATH, Huang CC, Lee WH, Yuan CC, Hsiao M, Deng L, Tzeng YM, Yeh CT. 4-Acetylantroquinonol B suppresses autophagic flux and improves cisplatin sensitivity in highly aggressive epithelial cancer through the PI3K/Akt/mTOR/p70S6K signaling pathway. Toxicol Appl Pharmacol. 2017 Jun 15; 325:48-60.
- Liu HW, Su YK, Bamodu OA, Hueng DY, Lee WH, Huang CC, Deng L, Hsiao M, Chien MH, Yeh CT, Lin CM. The disruption of the beta-catenin/TCF-1/STAT3 signaling axis by 4-acetylantroquinonol B inhibits the tumorigenesis and cancer stem-cell-like properties of glioblastoma cells, in vitro and in vivo. Cancers (Basel). 2018 Dec 5;10(12):491.
- Satriyo PB, Su CM, Huang WC, Fong IH, Aryandono T, Haryana SM, Deng L, Huang CC, Tzeng YM, Chao TY, Yeh CT*. 4-Acetylantroquinonol B Suppresses Cell Proliferation and Induces Apoptosis Through CDK2/CDK4-DDR Axis Inhibition in Triple Negative Breast Cancer. (Submitted).
STAY CONNECTED. SUBSCRIBE TO OUR NEWSLETTER.
Add your information below to receive daily updates.