Can Smoking Cause Breast Cancer?
Author(s)
Chiung-Tong ChenBiography
Chiung-Tong Chen, Ph.D., Investigator and Director, Institute of Biotechnology and Pharmaceutical Research, National Health Research Institutes (NHRI). Dr. Chen is a Ph.D. (1997) from the College of Pharmacy, Ohio State University, USA. He returned to join NHRI in 1999 to set up animal pharmacology and pharmacokinetic laboratories. Discovering drugs for unmet medical needs has been his core research interest. Equipped with multi-disciplinary expertise in pharmacology, pharmacokinetics, cancer biology, biopharmaceutical chemistry, toxicology and diseased animal models, his collaborative efforts for 21 years have led to 13 drug candidates being identified, 5 investigational new drug (IND) applications approved, and 3 Phase 1 studies implemented. He is a co-author of 139 peer-reviewed articles and co-inventor of 27 US/Taiwan/PCT patents.
Academy/University/Organization
National Health Research InstitutesSource
1.Artificial intelligence approach fighting COVID-19 with repurposing drugs. Biomed J. 2020, https://doi.org/10.1016/j.bj.2020.05.001 2.Repurposing old drugs as antiviral agents for coronaviruses. Biomed J. 2020, https://doi.org/10.1016/j.bj.2020.05.003-
TAGS
-
Share this article
You are free to share this article under the Attribution 4.0 International license
- LIFE SCIENCES
- Text & Image
- June 21,2020
The COVID-19 pandemic has caused more than 423,000 deaths. National Health Research Institutes (NHRI) planned and initiated the research fighting SARS-CoV-2-infected COVID-19 starting in January 2020, including repurposing existing drugs and discovering new drugs. In vitro assays for measurements of viral replication activities in feline infectious peritonitis virus, human coronavirus (HCoV-OC43), and SARS-CoV-2 were established and utilized to screen for the drugs/compounds reported to be active against SARS-CoV or SARS-CoV-2 and those suggested by an artificial intelligence approach. More than 20 drugs/compounds were found to be active against a coronavirus, and BPR3C0001 showed activity against SARS-CoV-2 replication with a high selective index. Tylophorines, which are purified from Tylophora Indica and have previously been shown to be active against cat, pig, murine, and human coronaviruses, including SARS-CoV, are here verified for effectiveness against SARS-CoV-2 replication in human cells. To further confirm their in vivo activities by using SARS-CoV-2-infected animals, e.g., ACE2 transgenic mice, is a must and is currently being prepared. We hope to identify a plant alkaloid for treating COVID-19. Furthermore, NHRI conducted a synthesis exercise of remdesivir, an investigational new drug developed by Gilead Sciences for treating Ebola virus infections. Remdesivir was clinically demonstrated to be effective in a COVID-19 patient in late January in the United States. The synthesis activity was initiated on February 5th and completed in 19 days with feasible synthetic schemes and high purity of grams-scale. Being always prepared in advance and being ready to deal with what healthcare issues could arise, NHRI has the missions to resolve national crises and to fulfill the emergency medical needs of Taiwan.
The National Health Research Institutes (NHRI) is a mission-oriented research organization. The Institute of Biotechnology and Pharmaceutical Research (IBPR) of NHRI aims to discover innovative therapeutics of novel small molecule and biologics for curing cancer, metabolic disorders, infectious diseases, neuronal disorders, aging, and others, including rare diseases. IBPR has been actively seeking collaboration opportunities with researchers in universities, hospitals, research institutes, and industries. IBPR aims to work with its industrial partners toward technology transfer out of the R&D achievements, with the hope of facilitating the development and improvement of the pharmaceutical and biotechnology industries in Taiwan. In response to the urgent national needs, NHRI engaged, in 2003 and 2005, research efforts to find 2 old drugs as anti-SARS agents and to synthesize oseltamivir for Tamiflu® manufacturing, respectively. At the beginning of the COVID-19 pandemic, discussions were initiated in January, and 4 discovery strategies were determined to find drugs for treating COVID-19, based on 22 years of experience in drug discovery and development of NHRI.
Strategy 1: Synthesis of remdesivir. Remdesivir is an investigational new drug developed by Gilead Sciences for treating Ebola virus infections and demonstrated active treating of a COVID-19 patient in January in the United States. It is a prodrug of nucleotide analogues (Figure 1) and inhibits RNA-dependent RNA polymerase. IBPR exercised its chemical synthesis in early February. Soon, within 22 days, remdesivir synthesis was completed in grams-scale of high-purity (>99%) with a recrystallization process established for scale-up synthesis and a technical report written. NHRI upholds the spirits of proactive planning, especially anti-epidemic deployment and being prepared in advance. The synthesis of remdesivir was meant to prepare the investigational drug beforehand only for possible emergency needs and to ease any possible concerns of the Taiwan people. A multi-center clinical study of remdesivir for COVID-19 therapy soon included Taiwan sites in early March. Following an Emergency Use Authorization in the United States and approval granted under an exceptional approval pathway in Japan as Veklury®, remdesivir was approved as a drug for treatment of COVID-19 in Taiwan in May, 2020.
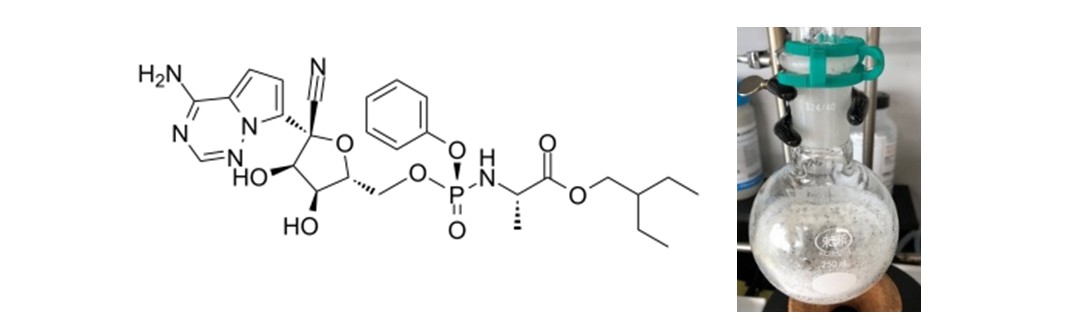
Figure 1: Chemical structure and recrystallized powder of remdesivir
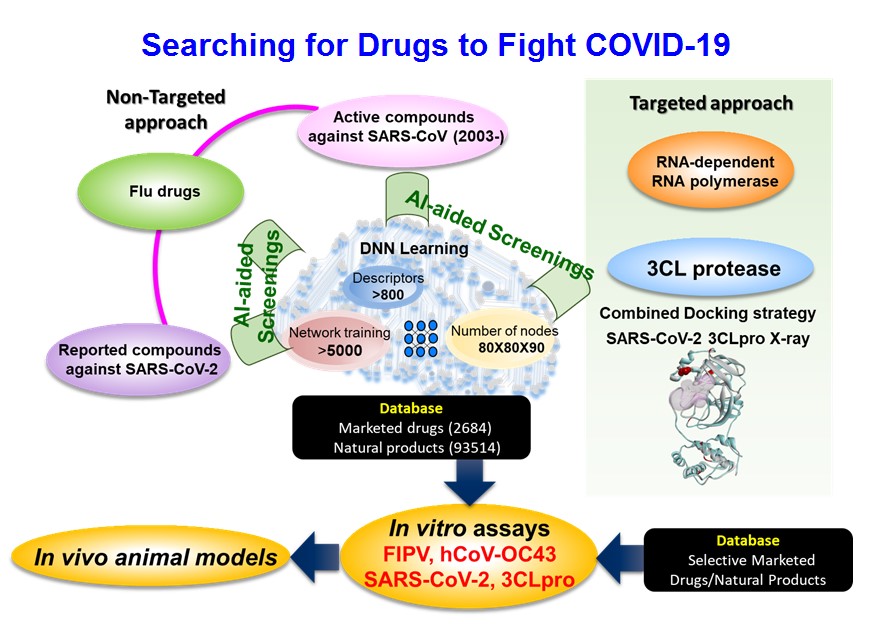
Strategy 2: Artificial Intelligence (AI) approach. AI techniques have been applied in drug discovery to offer quick and sometimes faithful predictions, and therefore have been adopted to search for old drugs and compounds that may inhibit SARS-CoV-2 replications. Two approaches: Non-Targeted, regardless of the biological molecular targets, and Targeted, on RNA-dependent RNA polymerase and 3CL protease were implemented. More than 900 molecular descriptors were used to describe the characteristics of biological activities and chemical structural details of those old drugs and compounds of small molecules being reported with promising activities against coronaviruses. Followed by reiterated deep learnings in the algorithm of deconvolutional neural network design, AI defined molecular descriptors set to identify AI-predicted old drugs and compounds out of the compounds databases (Figure 2). Eight AI-predicted old drugs and compounds were verified as being active against feline coronavirus replications. Further, AI implementations to find novel drugs as COVID-19 therapeutics are still ongoing.
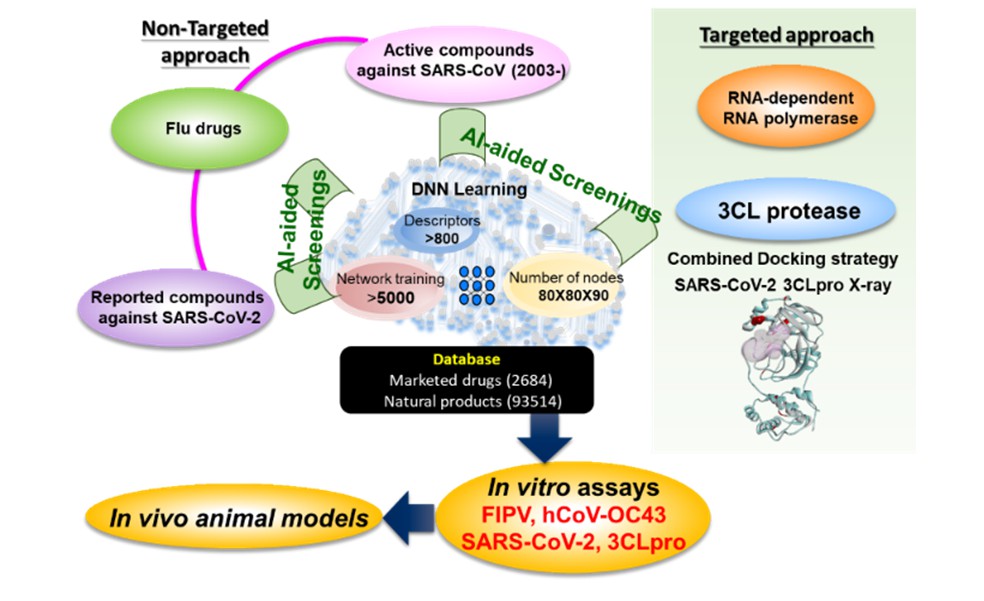
Figure 2: Non-Targeted and Targeted Approaches by Artificial Intelligence searching for promising old drugs and compounds
Strategy 3: Repurposing old drugs and compounds. In vitro assays using human coronavirus (HCoV-OC43/HCoV-229E), 3CL protease of SARS-CoV-2, and feline infectious peritonitis coronavirus (FIPV) in collaboration with the Agricultural Technology Research Institute have been established. Old drugs/compounds of small molecules being reported with promising activities against coronaviruses and those predicted by AI learning were collected and measured for antiviral activities in these 3 in vitro assays. More than 20 old drugs and compounds thus far showed inhibitory activity on virus replications, and their anti-SARS-CoV-2 activities are being examined. In collaboration with Professor Shih, Shin-Ru of the Chang Gung University, we found that a 3CL protease inhibitor BPR3C0001 (Figure 3) exhibited a potent neutralizing activity against SARS-CoV-2 (EC50=0.8 μM) and low cytotoxicity to the host cells (CC50>100 μM) with a high selective safety index of 114. Animal models of SARS-CoV-2 infection are being prepared to verify the efficacies of these old drugs and compounds, hoping to soon identify oral available, safe, and effective drugs for treating COVID-19.
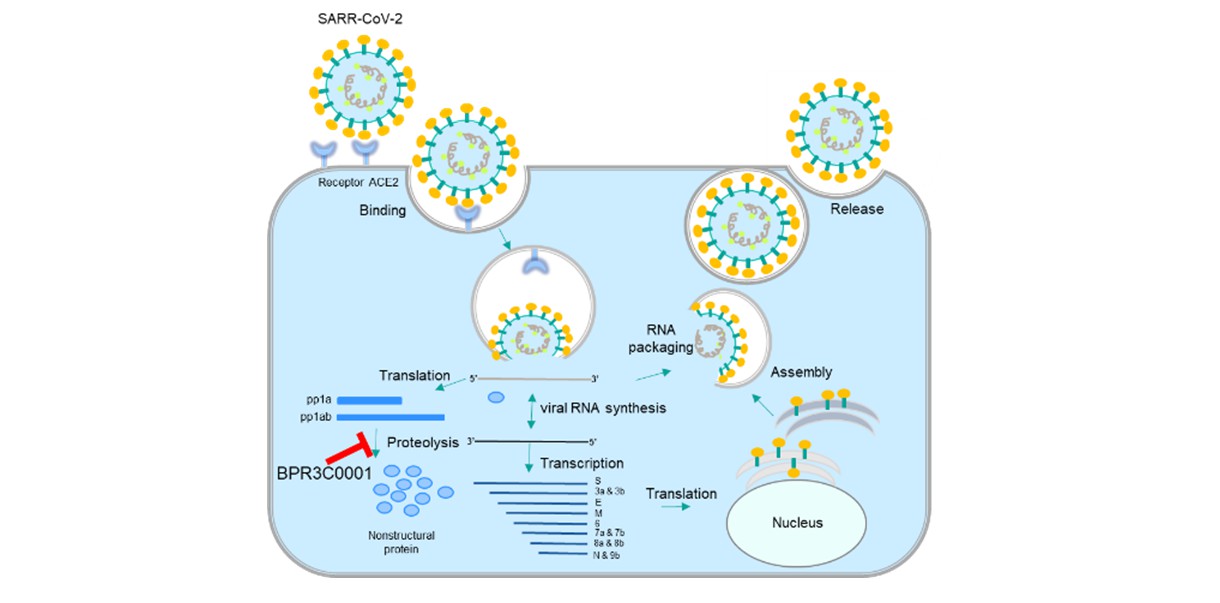
Figure 3: Inhibition of 3CL proteases by BPR3C0001
Strategy 4: Discovery of novel herb medicines. From our previous efforts searching for anti-SARS-CoV drugs during and after the SARS endemic in 2003, several tylophorine alkaloids were purified from Tylophorine Indica (Figure 4) and its derivatives had demonstrated broad-spectrum inhibition activities on swine transmissible gastroenteritis virus (TGEV), murine hepatitis virus (MHV), and human coronaviruses (SARS-CoV and HCoV-OC43). Tylophorine alkaloids are now again proven active against FIPV and human coronavirus (HCoV-OC43/HCoV-229E) with good in vitro potencies. A tylophorine derivative showed very potent inhibitory effects on SARS-CoV-2 (EC50=3-14 nM). In vivo models of SARS-CoV-2 infection using mice, e.g., ACE2 transgenic mouse model, and others are being prepared and will be established to verify the efficacies of these tylophorines. With the findings, we hope to identify a novel compound out of a medicinal herb showing clinical effectiveness for treating COVID-19 patients.

Figure 4: Photo of Tylophora Indica
STAY CONNECTED. SUBSCRIBE TO OUR NEWSLETTER.
Add your information below to receive daily updates.